Moderna’s novel coronavirus vaccine (mRNA-1273) is a front-running candidate to be one of the first available to market. If successful, the vaccine would also be the world’s first messenger RNA (mRNA) vaccine. Moderna has spent nine years developing mRNA technology that could theoretically manipulate the body to produce designer proteins with therapeutic or preventive benefits.
Moderna was quick to enter the race for a Covid-19 vaccine after reaching the clinical testing stage just 63 days after the genetic code of the virus was shared by Chinese scientists on January 11. In May, optimism swirled around the company after the release of promising partial phase 1 data sparked a rally, which has quadrupled the company’s valuation to over $30B.
According to consensus by Visible Alpha, Moderna’s Covid-19 vaccine revenue estimates span from around $100M to over $8B next year due to lack of agreement among analysts for pricing and usage. Many unknown factors complicate forecasts, such as an unknown number of future competing vaccines and usage within younger and older age cohorts.
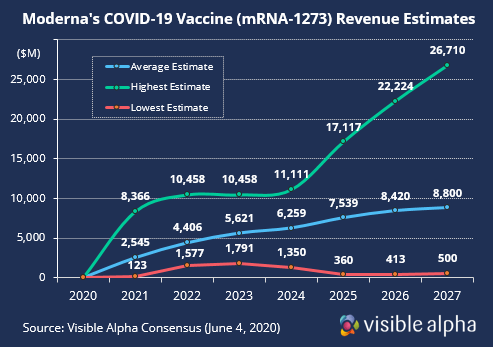
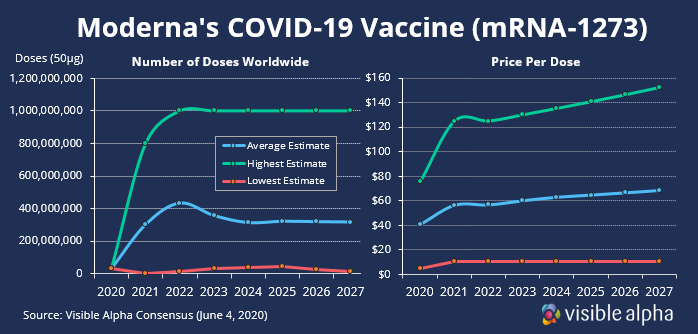
If phase 1 and 2 trials are successful, phase 3 could start as early as 3Q. And with the help of federal funding and a manufacturing agreement with Lonza, Moderna will have the capacity to produce millions of doses per year by the end of 2020 and a billion doses by 2021 if the vaccine is approved.
Learn more about the the impact of Covid-19 across every sector here.
This content was created using Visible Alpha Insights.
Visible Alpha Insights is an investment research technology platform that provides instant access to deep forecast data and unique analytics on thousands of companies across the globe. This granular consensus data is easily incorporated into the workflows of investment professionals, investor relations teams and the media to quickly understand the sell-side view on a company at a level of granularity, timeliness and interactivity that has never before been possible.